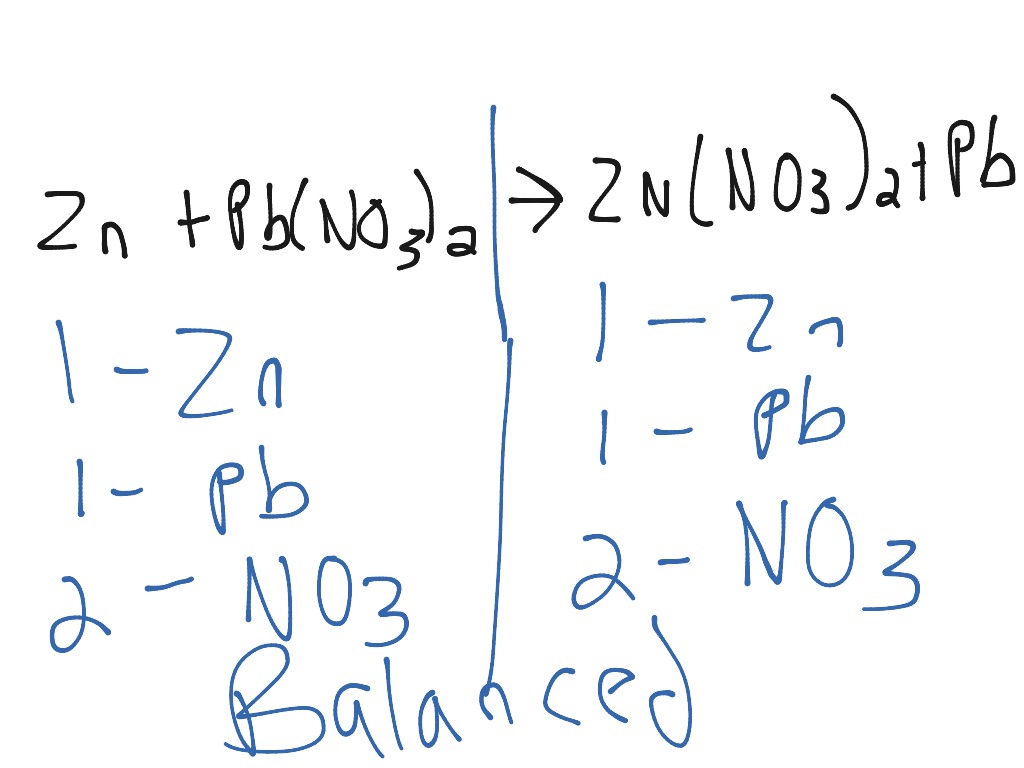
Post-16, students 'enjoy' this simple experiment as an introduction to electrochemistry. When teaching age 14-16 students about metallic bonding in the structure and bonding topic this demonstration can be used to illustrate the crystalline structure of metals. Watch our reactivity series of metals practical video to see metal displacement reactions set up in microscale (at 10:35) and find additional resources.See How to teach displacement reactions at 11–14 for more ideas and tips.(Note: 0.1 M lead nitrate is toxic.) Alternatively, you can do this in microscale using spotting tiles, which can be laid out in the same format as the table students use to record their results. Set up a series of test-tube reactions to investigate the displacement reactions between metals such as silver, lead, zinc, copper and magnesium and the salts ( eg sulfate, nitrate, chloride) of each of the other metals. Teaching goalsīy investigating a series of displacement reactions leaners aged 11–14 can learn about the reactivity series of metals. The three dimensional crystal structures will not stay in focus because their thickness varies and so constant focal adjustments are required. You will need to sink the copper before the reaction will work effectively. When you add the silver nitrate to the copper, the speck of copper can become suspended on the surface tension of the drop. If this is focused by a lens it can cause blindness. Take care not to reflect direct sunlight. If it is a sunny day, natural light can be used by positioning the microscope near a window. I switch off the light source and, on dark days, shine an LED, clamped in position using a retort stand and clamp, onto the sample from above to see the silver crystals in their true glory. Most microscopes have the light source positioned below the sample platform, which produces a dark outline of the crystals formed. The displacement reaction can be seen with the naked eye, but the crystalline structure of the silver is hard to make out. By using a microscope with an integrated digital camera still images can be recorded, printed and included in the students' notes.Īn alternative way to demonstrate this displacement reaction is to drop a 5 cm piece of 0.5 mm copper wire into a 8 cm depth of silver nitrate solution in a boiling tube. To show the demonstration to a class of students link a microscope to a view camera, or as I do, use a microscope with an integrated digital camera set at 40× magnification, linked to a laptop computer which is linked to a projector. Enough silver is produced at this magnification to fill the view within minutes. Crystals of silver will start to grow from the edges of the copper. Carefully add one drop of silver nitrate solution to the copper and refocus the microscope. (Note: for smaller microscopes a glass slide can be used instead of a petri dish.)įocus the microscope on the copper using ca 40× magnification (I use a 10× eye piece and 4× object lens). Using tweezers, put the speck on a petri dish and place the dish under a microscope. From this slice, cut off the smallest speck that you can see and handle, thus ensuring you have freshly cut edges which are not corroded. Using scissors, cut a very thin slice from the copper sheet. 
Silver nitrate solution is an irritant, wear eye protection and avoid skin contact.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
